At-a-glance
- 1 in 2 American children has a chronic disease.
- These disorders include, but not limited to food allergies, endocrine disorders, obesity, autoimmune diseases and neurocognitive disorders.
- The US – in common with many other nations – is faced with sick children, unhealthy food and a medical system unable to care for them.
- Disturbances in the epigenetic control of gene function and imbalances in gut bacterial populations (microbiome) are major contributing factors to these illnesses.
- Lifestyle interventions can help many sick children recover their health.
Recent studies confirm my clinical experience: 43% of children in the United States have a chronic illness, a percentage that rises to 52% when obesity is included as a disease.[1] In other words, a staggering 1 in 2 of American children now has a potentially lifelong disease. For the first time in modern history, children will be less healthy than their parents and will most likely live shorter lives.
Childhood diseases that are on the rise include food allergies, diabetes, inflammatory bowel disease, neurodevelopment disorders, celiac disease, non-celiac gluten sensitivity (NCGS), and obesity. Many of these diseases have multiple causes. For some diseases, there can be a time delay between the exposure and the disease actually showing up. For example, childhood cancers can take many years to develop.
It is clear from the steep rise in the incidence of these conditions (among many others not mentioned) over just a few decades that the causes are not primarily genetic, in terms of inheriting ‘faulty’ genes. It is known that some health problems are rooted in so-called “epigenetic changes”.
Epigenetics means “above genetics”. It is a system that consists of layers of different molecular structures that control the function or expression of the underlying genes encoded in the DNA. Thus epigenetics controls the overall pattern of gene function in a given organ or cell type within the body of an organism.
It is now known that lifestyle and environmental inputs can alter the epigenetic status within the organism, affecting the pattern of gene function for better or worse.
One type of epigenetic modification in particular – DNA methylation – becomes imprinted into DNA. This changes the genetic functioning of the organism that was initially affected. In addition, certain DNA methylation patterns, newly acquired during the lifetime of the parent, can be passed down to their children and even to further generations.
Importantly, epigenetics is a dynamic system. Inappropriate lifestyle habits, including poor diet and exposure to environmental stressors such as chemical pollutants, can result in epigenetic changes, leading to disturbances in gene expression. This in turn can result in ill health. But conversely, positive lifestyle changes can reset the epigenetic and gene function profile, restoring the individual to good health.[2],[3]
Another important system that is now known to be crucial to good health is balance in the bacterial population of the digestive system (the gut microbiome). Imbalance (“dysbiosis”) in the gut microbiome has been shown to contribute to a plethora of ill health conditions, including obesity, type 1 diabetes, autoimmunity (where the immune attacks healthy cells and tissues), mood disorders, nerve degeneration, and heart disease.[4] Fortunately, as with epigenetic status, the gut microbiome is a dynamic system. If it has entered into a state of dysbiosis, for example, through poor diet, it can be brought back to a balanced state, leading to amelioration of a disease condition.
It is fascinating, and perhaps not surprising, that the two systems of epigenetic status of different organs and systems of the body and the gut microbiome have been found to be intimately linked, with molecules produced in the gut influencing gene expression in the body through changes in epigenetic control mechanisms.[5]
This understanding of epigenetics and the importance of maintaining gut microbiome balance provide mechanisms that could explain my clinical experience gained over almost 40 years – which shows that lifestyle changes can result in rapid and significant improvement in many conditions. I’ll return to that topic at the end of this article.
Following is an overview of the major health conditions that are rapidly escalating in our children.
1. Food allergies
Results from a 2009–2010 survey of more than 38,000 children indicate that 5.9 million children, or 8%, have food allergies.[6] That’s 1 in 13 children, or roughly two in every classroom. And food allergies are on the rise. The Centers for Disease Control and Prevention (CDC) report that between 1997 and 2011, food allergy prevalence among children increased by 50%.[7] Hospitalizations for severe food allergies have increased substantially over the last two decades.[8]
These data only take into account severe food allergies. They do not include the low-grade sensitivities or intolerances to dairy, soy, and legumes, which now affect vast numbers of children. The latter are under-reported due to the fact that they are not commonly evaluated in pediatric practice.
No one can enter a classroom now without being made aware of food warnings and restrictions. In California, a 2014 law requires that every school site must have an Epipen anti-allergy injector to treat potentially life-threatening reactions to food (anaphylaxis).[9]
An increasing number of studies suggest that epigenetic regulation of gene expression plays a critical role in the development of allergic diseases and that this regulation can be affected by diet.[10] Other studies support a link between gut microbiome dysbiosis and food allergy.[11]
2. Diabetes
Type 1 diabetes is a serious lifelong condition in which the blood glucose level is too high because the pancreas produces little or no insulin, a hormone that allows uptake of glucose by the body’s cells to produce energy from food eaten or to store the glucose for future use. The rate of this disease has been rising in recent decades, both in the US and in Europe. Unlike other diseases, Type 1 diabetes is easy to correctly diagnose and hard to miss. Left untreated, the disease is fatal.
In 2015, 193,000 children and adolescents younger than age 20 (0.24% of the total US population younger than age 20) were reported to have diabetes.[12] This number does not include pre-diabetes, a condition that is linked to obesity and is rising alarmingly in children. Nor does it include the vast number of children with Type 2 diabetes and/or metabolic syndrome. Type 2 diabetes is a serious condition in which the insulin made by the pancreas is either not used properly by the body (“insulin resistance”) or the pancreas cannot make enough insulin. Metabolic syndrome is a cluster of conditions – including increased blood pressure, high blood sugar, excess body fat around the waist, and abnormal blood cholesterol or triglyceride levels — that occur together, increasing the risk of heart disease, stroke, and diabetes.
The rise in childhood diabetes began in the mid-20th century. The best US data available to examine trends comes from the Erie County study set up in 1962. The study reports an incidence of 11.3 per 100,000 people per year in 1959–1961. But by the end of the 20th century, some 3–4 children out of 1,000 in Western industrialized countries would require insulin treatment, and a steady rise in incidence had been reported from many other parts of the world.[13]
Between 2002 and 2012, the rate of newly diagnosed cases of type 1 diabetes in youth in the US increased by about 1.8 percent each year. During the same time period, the rate of new diagnosed cases of type 2 diabetes increased even more quickly, at 4.8 percent each year.[14],[15]
Epigenetic changes leading to alterations in gene function are involved in the inflammatory aspects of type 2 diabetes, and inflammation plays an important role in all aspects of this illness. Therefore researchers are looking at ways of modifying the epigenetic status in order to cure or prevent the disease.[16]
Allergenic foods and gut microbiome dysbiosis are suspected triggers for type 1 diabetes. Gut dysbiosis seems to increase intestinal permeability (causing “leaky gut” syndrome), which in turn leads to inflammation and autoimmunity. Encouraging a healthier gut microbiome appears to be a promising preventative method and therapy for this disease.[17] Gut microbiome dysbiosis has been found to be a cause of type 2 diabetes and metabolic syndrome.[18]
3. Inflammatory bowel disease (IBD)
Autoimmune bowel diseases have been increasing in children, most notably since 1990. Two of these diseases, ulcerative colitis and Crohn’s disease, are distinctly on the rise. This group of diseases is often life-altering for children and can lead to the development of other serious disorders, such as cancers, later in life. Canada has the highest reported rate of IBD. The Canadian data show that both the prevalence (commonness) and incidence (rate of newly diagnosed cases) of IBD have increased remarkably from 1994 to 2005, especially in younger children.[19] These trends are comparable to worldwide patterns.
There is growing evidence that epigenetic factors, such as DNA methylation, play a role in the development of IBD and that DNA methylation in the gut can react to changes in the microbiota.[20]
Current scientific opinion holds that genetically predisposed people develop gut microbiome dysbiosis and that inflammation develops as a result of environmental stressors. Genetic predisposition or dysbiosis in isolation do not lead to IBD. Instead, complex interactions between the environment, genetics, gut microbiota, and the immune system appear to be responsible.[21]
4. Neurodevelopmental and behavioral disorders
The rates of attention deficit hyperactivity disorder (ADHD), autism spectrum disorder (ASD), and neurodevelopmental disability have been increasing exponentially within the last two decades.[22] Previously this rise was attributed to improvements in our ability to detect these problems. However, this explanation has fallen by the wayside as the rates of such disabilities in children are skyrocketing. Exposure to certain pesticides during pregnancy is among known risk factors for neurodevelopmental disorders, including lower IQ and impaired motor skills.[23]
A study published in the Journal of the American Medical Association (JAMA) looked at the prevalence of ADHD in US children and teenagers over a 20-year period. During the period 1997–1998, the prevalence of ADHD was 6.1%. But this number increased to 10.2% by 2015–2016. All subgroups by age, sex, ethnicity, family income, and geographic region showed a statistically significant increase in prevalence.[24]
The National Health Center for Health Statistics released a report on developmental disability in children, including autism. The report found that in the two years between 2014 and 2016 – an extremely short time frame – the prevalence of developmental disability in children aged 3–17 years increased from 5.76 to 6.99% (see graph below), or 1 in 14 children.[25]
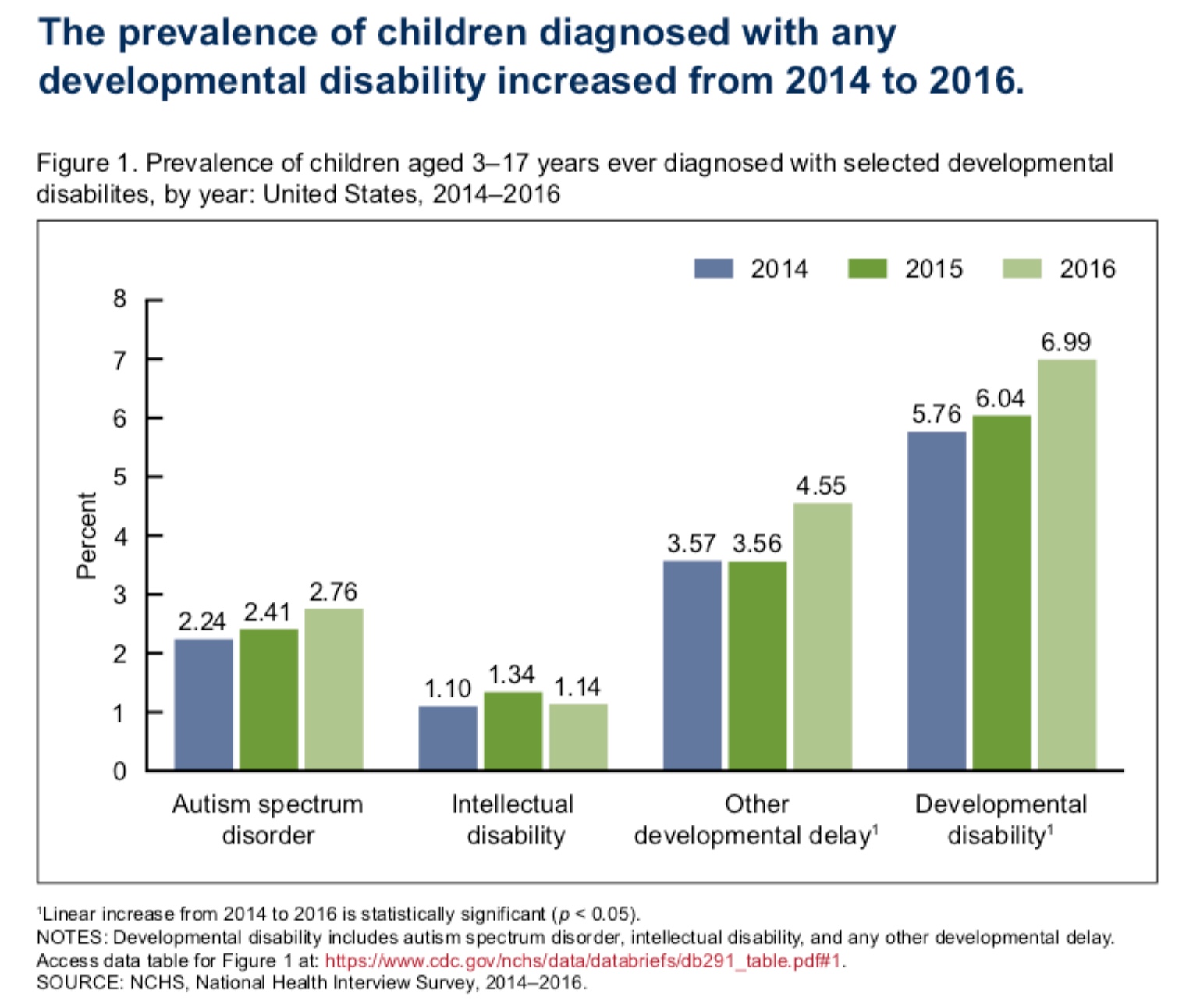
In 2014, ASD affected 1 in 59 children and was four times as common in boys as in girls.[26] This qualifies as an epidemic. According to data from the US CDC, there are wide differences between states, with New Jersey having the highest rate. The CDC states that it is unknown to what extent environmental or genetic factors play a role.[27] However, the rapid rise in this condition means that even if genetic factors do play a role, there are also strong environmental triggers at work.
Epigenetics is believed to play a role in the development of ASD, though research is ongoing to identify which epigenetic changes are associated with the disorder and which environmental triggers may be at work.[28]
Many studies show that an abnormal gut microbiome is related to ASD and that treatments that alter the gut microbiome result in improvements in ASD symptoms.[29]
5. Celiac disease and non-celiac gluten sensitivity
Celiac disease (CD) is a genetically predisposed autoimmune disease that is being diagnosed with increased frequency in both children and adults.[30] ,[31] Another condition, non-celiac gluten sensitivity (NCGS), also known as non-celiac wheat sensitivity (NCWS), is also on the rise. The causes and triggers of both diseases are poorly understood.[32]
The prevalence of CD in children in the Denver area has been estimated at up to 3% by the age of 15.[33]
Almost all patients diagnosed with CD have variations in at least two of the genes that play a role in enabling the organism to distinguish between self and non-self (known as the “major histocompatibility complex” genes). But while 40% of people have these gene variants, not everyone who has them goes on to develop CD.[32] This suggests that either variations in other genes or environmental factors, or both are the cause of this disease.
Thus the rise in prevalence of CD cannot be fully explained by genetic factors. Celiac researcher and gastroenterologist Edwin Liu, of the University of Colorado School of Medicine, explained, “Over decades, it’s just too quick for genetic changes to occur. We have to assume that this is based on environmental factors.”[32] It is not known which environmental factors are to blame.
Nor can the rise in prevalence be attributed to improvements in diagnosis. Joseph Murray, a celiac researcher at the Mayo Clinic in Rochester, Minnesota, commented that data indicating rising rates in the US showed that “We weren’t just better at finding celiac disease”, but that “There was a lot more of it to go around.”[32]
This trend began before the introduction in 1996 of genetically engineered food and the associated increases in pesticide use, but has continued to rise since then.
A disturbance in epigenetic regulation of expression of certain genes has been found to be involved in CD.[34] Marked gut microbiome dysbiosis has also been found in the small intestine of adults with CD and this bacterial imbalance is suspected of contributing to the inflammation that characterizes the disease.[35]
6. Obesity
In the US, the percentage of children and teens with obesity has more than tripled since the 1970s.[36] Data from 2015–16 show that nearly 1 in 5 children aged 6-19 years old is obese.[37]
Obesity has serious knock-on effects on the physical, emotional and mental well-being of children. Obese children are at higher risk for other health conditions such as type 2 diabetes, heart disease, asthma, sleep apnea, and musculoskeletal conditions. They are apt to be bullied at school and suffer from mental health issues due to social marginalization and low self-esteem.[38] Obese children will likely go on to become obese adults, carrying with them the health risks mentioned above, as well as an increased risk of colorectal cancer for males.[39]
The obesity epidemic is not primarily due to genetics but to lifestyle and environmental factors. However, researchers believe it likely that obesity is associated with both genetic and environmental influences on epigenetic status.[40]
Recent studies suggest that obesity is associated with gut microbiome dysbiosis. Obese people have been found to have less intestinal bacterial diversity.[41]
Case studies
The US – in common with many other developed nations – is faced with sick children, unhealthy food, and a medical system unable to care for them.[42] Fortunately, lifestyle interventions can make a dramatic difference to children’s health. In my clinical experience, the most successful intervention is switching the whole family to a diet based on organically grown foods that are as little processed as possible and cooked fresh at home.
For example, 5-year-old Chase was a boy who was brought to me because of his food allergy symptoms, including recurrent abdominal pain, constipation and eczema. He was having trouble starting kindergarten because of his discomfort. Upon evaluation, Chase was found to have both IgE and IgG antibodies (which the body produces in response to foreign “threats”) to dairy and gluten. The family ate organic food inconsistently. Upon switching to organic food and slowly phasing out the food culprits (this transition was difficult for the family since they believed he would become calcium-deficient without milk), Chase’s health issues resolved within a month.
Many of my child patients have been diagnosed with ASD. This generally means frequent clinic visits since these children have many associated disorders such as gastrointestinal complaints, sleep disruptions, behavioral issues, and infections. A large proportion of children “on the spectrum” that I’ve cared for have had issues with gluten, dairy, and occasionally soy. These children may also be picky eaters, so removing the junk food from their diet can be challenging. Many parents lament that this is all they can get their child to eat. This was the case with Juan, a 6-year-old boy from the Central Valley of California. But his family persisted with changing his diet to organic and later eliminating allergenic foods. The switch resulted in a complete resolution of his gastrointestinal complaints – as well as improving his mood.
While such a dietary change is just a first step in the treatment, parents report their children having less intestinal complaints, improved sleep, and better behavior.
These case examples and many more exemplify the tenet of the ancient Greek physician Hippocrates,who said, “Let food be thy medicine”. We can update this for our current times to say, “Let organic food be thy epigenetic and gut microbiome medicine.” Such a diet lays the foundation of good health for children and adults alike.
References
- Bethell CD, Logan MD, Strickland BB, Schor EL, Robertson J, Newacheck PW. A national and state profile of leading health problems and health care quality for US children: key insurance disparities and across-state variations. Acad Pediatr. 2011 May-June; 11(3 Suppl):S22-23.↑
- Sagner M et al (2014). Lifestyle medicine potential for reversing a world of chronic disease epidemics: from cell to community. Int J Clin Pract. 2014 Nov;68(11):1289-92. doi: 10.1111/ijcp.12509. ↑
- Feinberg AP (2018). The key role of epigenetics in human disease prevention and mitigation. N Engl J Med 378:1323-1334. ↑
- Kho ZY (2018). The human gut microbiome – a potential controller of wellness and disease. Front Microbiol. 9:1835. doi: 10.3389/fmicb.2018.01835. ↑
- Qin Y (2018). Crosstalk between the microbiome and epigenome: messages from bugs. J Biochem. 163(2):105-112. doi: 10.1093/jb/mvx080. ↑
- Gupta RS, Springston MR, Warrier BS, Rajesh K, Pongracic J, Holl JL (2011). The prevalence, severity, and distribution of childhood food allergy in the United States. Pediatrics 128(1):e9-17. ↑
- Jackson KD, Howie LD, Akinbami LJ (2013). Trends in allergic conditions among children: United States, 1997-2011. NCHS Data Brief No 121. Hyattsville, MD: National Center for Health Statistics. http://www.cdc.gov/nchs/products/databriefs/db121.htm ↑
- Lin RY, Anderson AS, Shah SN, Nurruzzaman F (2008). Increasing anaphylaxis hospitalizations in the first two decades of life: New York State, 1990-2006. Ann Allergy Asthma Immunol 101(4):387-93. ↑
- California Legislature (2014). Senate Bill No. 1266, Chapter 321. http://leginfo.legislature.ca.gov/faces/billNavClient.xhtml?bill_id=201320140SB1266 ↑
- Lee SE, Kim H (2016). Update on early nutrition and food allergy in children. Yonsei Med J. 57(3):542–548. doi: 10.3349/ymj.2016.57.3.542. ↑
- Blázquez AB, Berin C (2017). Microbiome and food allergy. Translational Research 179: 199-203. ↑
- American Diabetes Association (2018). Statistics about diabetes: Overall numbers, diabetes and prediabetes. http://www.diabetes.org/diabetes-basics/statistics/ ↑
- Gale EAM (2002). The rise of childhood type 1 diabetes in the 20th century. Diabetes 51(12): 3353-3361. http://diabetes.diabetesjournals.org/content/51/12/3353 ↑
- National Institutes of Health (2017). Rates of new diagnosed cases of type 1 and type 2 diabetes on the rise among children, teens. April 13. https://www.nih.gov/news-events/news-releases/rates-new-diagnosed-cases-type-1-type-2-diabetes-rise-among-children-teens ↑
- Mayer-Davis EJ et al (2017). Incidence trends of type 1 and type 2 diabetes among youths, 2002–2012. N Engl J Med 2017(376):1419-1429. https://www.nejm.org/doi/full/10.1056/NEJMoa1610187 ↑
- Sommese L et al (2017). Clinical relevance of epigenetics in the onset and management of type 2 diabetes mellitus. Epigenetics 12(6): 401–415. doi: 10.1080/15592294.2016.1278097 ↑
- Bibbò S et al (2017). Is there a role for gut microbiota in type 1 diabetes pathogenesis? Ann Med. 49(1):11-22. doi: 10.1080/07853890.2016.1222449. ↑
- Li x et al (2017). Gut microbiota dysbiosis drives and implies novel therapeutic strategies for diabetes mellitus and related metabolic diseases. Front Immunol. 8:1882. doi: 10.3389/fimmu.2017.01882. ↑
- Benchimol EI et al (2017). Trends in epidemiology of pediatric inflammatory bowel disease in Canada: Distributed network analysis of multiple population-based provincial health administrative databases. The American Journal of Gastroenterology 112: 1120–1134. https://www.nature.com/articles/ajg201797 ↑
- Kellermayer R (2012). Epigenetics and the developmental origins of inflammatory bowel diseases. Can J Gastroenterol. 26(12): 909–915. ↑
- Abegunde AT et al (2016). Environmental risk factors for inflammatory bowel diseases: Evidence based literature review. World J Gastroenterol. 22(27):6296–6317. ↑
- Halfon N et al (2012). The changing landscape of disability in childhood. The Future of Children 22(1):13-42. https://www.ncbi.nlm.nih.gov/pubmed/22550684 ↑
- Shelton JF et al (2014). Neurodevelopmental disorders and prenatal residential proximity to agricultural pesticides: The CHARGE Study. Environmental Health Perspectives 122(10): 1103–1110. https://ehp.niehs.nih.gov/doi/10.1289/ehp.1307044 ↑
- Xu G et al (2018). Twenty-year trends in diagnosed attention-deficit/hyperactivity disorder among US children and adolescents, 1997-2016. JAMA Network Open 1(4):e181471. doi:10.1001/jamanetworkopen.2018.1471 ↑
- NCHS (2017). Estimated prevalence of children with diagnosed developmental disabilities in the United States, 2014–2016. NCHS Data Brief No 291. November. https://www.cdc.gov/nchs/data/databriefs/db291.pdf ↑
- Centers for Disease Control and Prevention (2018). Autism spectrum disorder (ASD): Data and statistics. https://www.cdc.gov/ncbddd/autism/data.html ↑
- Centers for Disease Control and Prevention (2014). Community report on autism. https://www.cdc.gov/ncbddd/autism/states/comm_report_autism_2014.pdf ↑
- Siu MT, Weksberg R (2017). Epigenetics of autism spectrum disorder. In: Delgado-Morales R. (eds) Neuroepigenomics in Aging and Disease. Advances in Experimental Medicine and Biology, vol 978. Springer, Cham. ↑
- Li Q et al (2017). The gut microbiota and autism spectrum disorders. Front Cell Neurosci. 11:120. doi: 10.3389/fncel.2017.00120. ↑
- Ludvigsson JF et al (2013). Increasing incidence of celiac disease in a North American population. Am J Gastroenterol 108(5): 818–824. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3686116/ ↑
- Liu E et al (2017). High incidence of celiac disease in a long-term study of adolescents with susceptibility genotypes. Gastroenterology 152(6):1329-1336.e1. doi: 10.1053/j.gastro.2017.02.002 ↑
- Offord C (2017). The celiac surge. The Scientist, June 1. https://www.the-scientist.com/features/the-celiac-surge-31438 ↑
- Liu E et al (2017). High incidence of celiac disease in a long-term study of adolescents with susceptibility genotypes. Gastroenterology 152(6):1329-1336.e1. doi: 10.1053/j.gastro.2017.02.002 ↑
- Oittinen M et al (2016). Polycomb repressive complex 2 enacts Wnt signaling in intestinal homeostasis and contributes to the instigation of stemness in diseases entailing epithelial hyperplasia or neoplasia. Stem Cells 35(2):445–457. ↑
- D’Argenio V et al (2016). Metagenomics reveals dysbiosis and a potentially pathogenic N. flavescens strain in duodenum of adult celiac patients. Am J Gastroenterol. 2016 Jun; 111(6): 879–890. doi: 10.1038/ajg.2016.95. ↑
- Fryar CD et al (2016). Prevalence of overweight and obesity among children and adolescents aged 2–19 years: United States, 1963–1965 through 2013–2014. NCHS Health E-Stats. July. https://www.cdc.gov/nchs/data/hestat/obesity_child_13_14/obesity_child_13_14.pdf ↑
- Hales CM et al (2017). Prevalence of obesity among adults and youth: United States, 2015–2016. NCHS Data Brief No 288. October. https://www.cdc.gov/nchs/data/databriefs/db288.pdf ↑
- Pulgarón ER (2013). Childhood obesity: A review of increased risk for physical and psychological co-morbidities. Clin Ther 35(1): A18–A32. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3645868/ ↑
- Maffeis C, Tatò L (2001). Long-term effects of childhood obesity on morbidity and mortality. Horm Res 55 Suppl 1:42-5 ↑
- Herrera BM et al (2011). Genetics and epigenetics of obesity. Maturitas. 69(1):41–49. doi: 10.1016/j.maturitas.2011.02.018. ↑
- DeGruttola AK et al (2016). Current understanding of dysbiosis in disease in human and animal models. Inflamm Bowel Dis. 22(5):1137–1150. doi: 10.1097/MIB.0000000000000750. ↑
- Perro M and Adams V (2017). What’s Making Our Children Sick? How Industrial Food Is Causing an Epidemic of Chronic Illness, and What Parents (and Doctors) Can Do About It. Chelsea Green. https://www.chelseagreen.com/product/whats-making-our-children-sick/ ↑

